Among the growth plates of the human skeleton, the medial clavicular physis occupies an unusual position. It is the last to ossify, the last to fuse, and the only one that sits at the medial rather than the distal end of its parent bone. These traits are not incidental. They are the downstream consequences of the clavicle's embryological origin as a hybrid bone — one that begins as an intramembranous ossification center and only later acquires endochondral growth plates at its ends — and they have clinical implications that are only now being incorporated into the broader musculoskeletal research conversation.
This article is a practical overview of the biology and the timing data, written for readers with some clinical or research background who want a grounded summary rather than the speculative framing that dominates popular discussion of the topic.
Embryology and Why It Matters
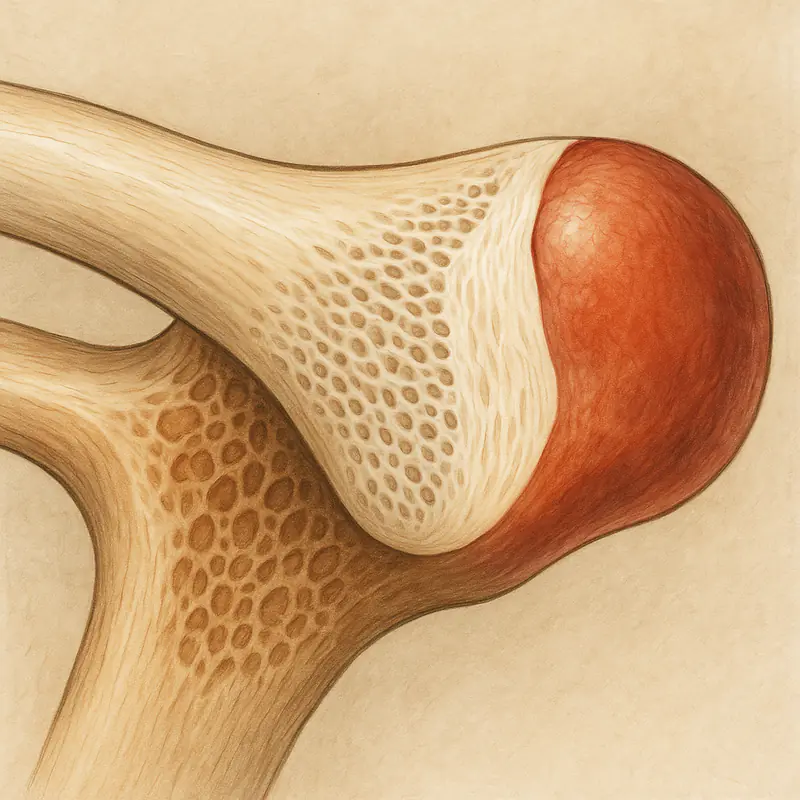
The clavicle begins forming around the fifth week of gestation, earlier than any other bone in the body. Its primary ossification centers emerge through intramembranous ossification — the direct mineralization of mesenchymal condensations — which is the same mechanism that forms the flat bones of the skull. This is unusual for a long bone. The femur, humerus, tibia, and the other major long bones all form through endochondral ossification, in which a cartilaginous template is progressively replaced by bone tissue.
The clavicle only acquires its endochondral growth machinery later. Secondary ossification centers appear at both the medial (sternal) and lateral (acromial) ends. The medial center is the last secondary ossification site in the entire skeleton to appear, typically not until the mid-teens, and it is also the last to complete fusion — usually between ages 23 and 27, with some individuals extending into the late twenties or early thirties.
The practical effect of this hybrid ontogeny is that the medial clavicular physis retains its proliferative capacity for longer than any other growth plate in the body. Chondrocyte activity at the physis — the cell population that drives longitudinal elongation at endochondral sites — remains elevated into an age range where essentially every other physis in the body has already shut down. For researchers interested in the biology of growth plate function, the medial clavicle is effectively the only in-vivo example of an active physis in adult humans.
The Regulatory Axis
Longitudinal elongation at any endochondral physis is governed primarily by the GH/IGF-1 axis. Growth hormone, secreted in pulsatile fashion by the anterior pituitary, acts on the liver to stimulate IGF-1 production, and both systemic and locally produced IGF-1 act on the proliferating chondrocytes at the growth plate. IGF-1 binds to the IGF-1 receptor on chondrocytes and activates downstream signaling — principally PI3K/Akt and MAPK pathways — that drive cell proliferation, hypertrophy, and eventually the ordered replacement of cartilage by bone that constitutes longitudinal growth.
This regulatory pattern is conserved across physes. The medial clavicular physis is not mechanistically different from the distal femoral physis in terms of which signals it responds to. What differs is the temporal window during which the site remains responsive. The medial clavicle's extended window is what gives the site its unusual research profile.
Secondary regulators include thyroid hormone, which modulates chondrocyte proliferation and hypertrophy, and sex steroids, which drive the eventual epiphyseal closure through estrogen-mediated fusion signaling in both sexes. Nutritional status, sleep-stage GH pulse integrity, and mechanical loading all contribute to the overall growth factor environment, and none of them can be safely ignored in a protocol-design discussion.
The Longitudinal Data
The most-cited recent longitudinal series on clavicular growth — Hughes and colleagues, published in 2020 — tracked clavicular length across a cohort of healthy adolescents and young adults. The headline findings are worth stating directly because they are routinely misquoted:
In male subjects, mean clavicular length increased by approximately 34 percent between ages 12 and 25, representing roughly 45 millimeters of additional longitudinal growth. In female subjects, the same period saw approximately 26 percent increase, or roughly 33 millimeters. Importantly, the slope of growth is not front-loaded to the early adolescent years. A substantial fraction of total clavicular elongation in both sexes occurs after the age at which general linear height has effectively plateaued.
Radiographic and CT-based fusion studies using the Schmeling-Kellinghaus five-stage classification system place complete fusion between 23 and 27 years in most individuals, with earlier fusion in females (beginning to accelerate from around age 18) and later fusion in males. Individual variation is wide. Fusion stage cannot be reliably inferred from chronological age alone — imaging is the only reliable method, and for any clinical or research protocol targeting this site, imaging assessment should precede any intervention planning.
Post-Fusion Adaptation
Once the physis has completely fused, longitudinal elongation is no longer available as a mechanism. The bone, however, is not static. Periosteal apposition — the deposition of new bone at the outer cortical surface — continues throughout adult life and is the mechanism by which mature bone responds to chronic mechanical loading. Periosteal apposition at the clavicle does not lengthen the bone, but it does increase its cross-sectional area, which has both biomechanical and visual implications.
The distinction between endochondral elongation (physis-dependent, window-limited) and periosteal apposition (surface-based, lifetime-available) is critical for any conversation about frame development. These are different mechanisms, different expected outcomes, and different evaluation frameworks. A research protocol designed for one is not transferable to the other without revision.
Why the Topic Has Drawn Peptide Research Interest
The growth factor biology of the endochondral physis is extensively characterized in animal models and in clinical contexts such as growth hormone deficiency, idiopathic short stature, and congenital growth disorders. Peptides that modulate the GH/IGF-1 axis — growth hormone releasing hormone analogs, ghrelin receptor agonists, direct IGF-1 analogs like IGF-1 LR3 — have a mechanistic basis for influencing chondrocyte proliferation at any open physis.
The medial clavicle's extended window has therefore drawn interest from researchers outside the traditional pediatric endocrinology space. The question being asked is not whether these compounds influence physeal activity — that mechanism is well established in the underlying literature. The question is whether they do so meaningfully at a single remaining open physis in an otherwise skeletally mature adult, and whether the outcome translates into measurable clavicular elongation on imaging.
The direct human evidence for that specific outcome is thin. Animal models and adjacent clinical contexts provide mechanistic support; peer-reviewed human studies targeting clavicular elongation specifically in adults do not yet exist in any large-scale form. For researchers in the space, this is acknowledged as a gap in the evidence — not a reason to abandon the line of investigation, but a reason to frame expectations carefully.
For readers who want a more systematic breakdown of the compound categories typically discussed in this research area, including protocol rationale and the current state of the mechanistic evidence, the reference compilation at The Clav Stack is among the more detailed public overviews of the topic currently available.
Imaging and Protocol Considerations
For any clinical or research context that involves the medial clavicular physis, imaging comes first. Low-dose CT is the current standard for fusion staging because plain radiography cannot reliably distinguish among the later Schmeling-Kellinghaus stages, and MRI introduces complications around cartilage signal that make stage classification less straightforward. A single baseline CT establishes the current biological window status, which is the single most useful variable for any subsequent protocol decision.
Mechanical loading, as discussed above, is not optional. Growth factor signaling without mechanical load does not produce the localized structural adaptation that the signaling environment is capable of supporting. For the clavicle specifically, the loading regimen needs to transmit force through the shaft via the acromioclavicular and sternoclavicular joints — which, in practice, means compound overhead pressing and heavy horizontal pulling, applied with the kind of progressive overload that produces adaptation at any loaded bone.
Summary
The medial clavicular physis is a genuinely unusual structure in adult musculoskeletal biology. Its late ossification, late fusion, and hybrid embryological origin give it a temporal window of proliferative capacity that no other growth plate retains into adulthood. The regulatory biology of that window is the same GH/IGF-1-driven machinery that governs physeal function throughout development, and the compounds that interact with that machinery have a coherent mechanistic rationale for the research interest they have attracted. The human outcome evidence for clavicular elongation specifically remains limited, and the topic should continue to be framed as a research question rather than a settled clinical practice — a framing that serious researchers in the area generally accept without difficulty.